Immunomodulator Withdrawal From Anti-TNF Therapy Is Not Associated With Loss of Response in Inflammatory Bowel Disease - Clinical Gastroenterology and Hepatology

IJMS | Free Full-Text | The Role of TNF-α and Anti-TNF-α Agents during Preconception, Pregnancy, and Breastfeeding
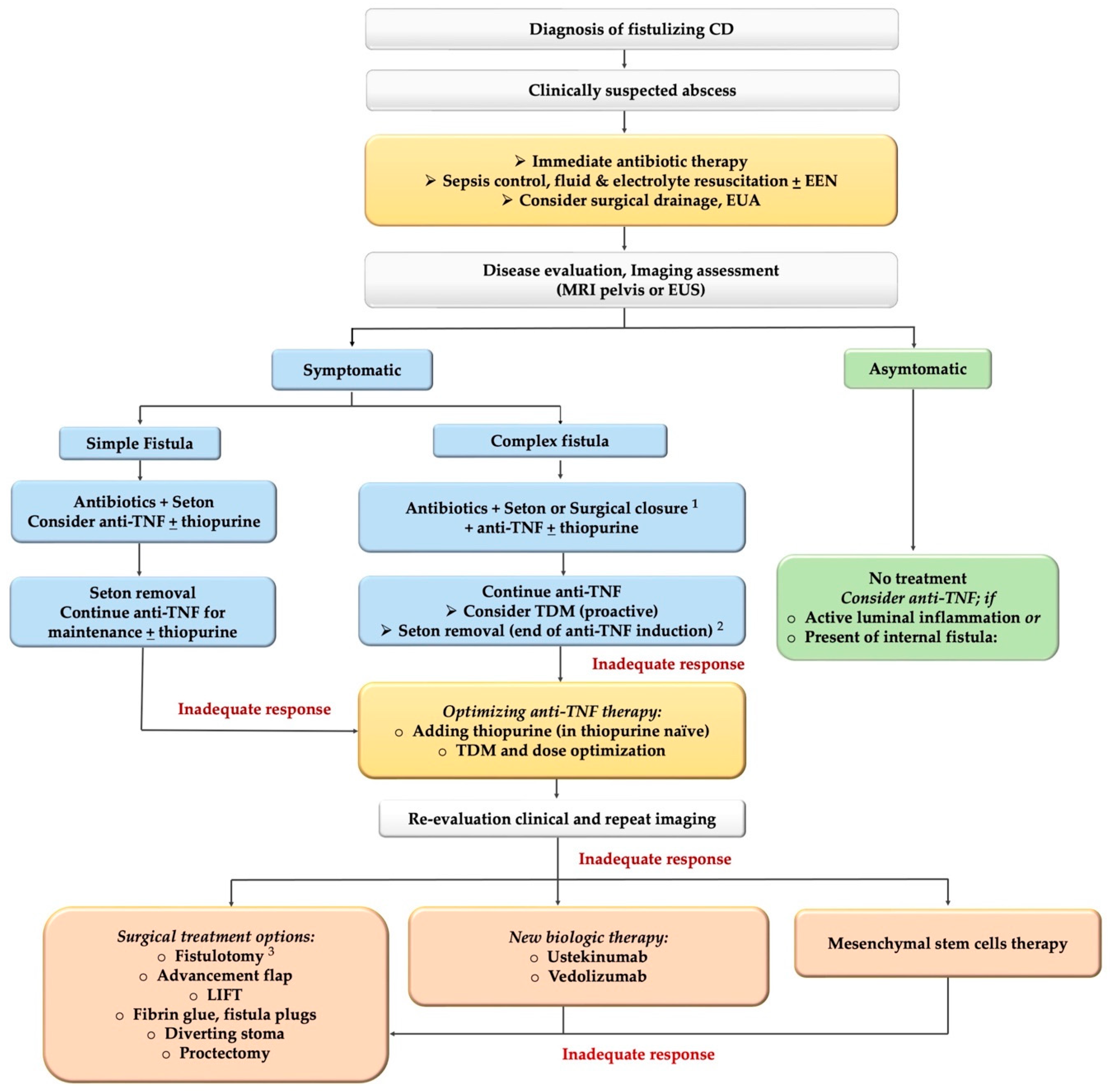
Treatment Algorithms for Crohn's Disease - FullText - Digestion 2020, Vol. 101, Suppl. 1 - Karger Publishers
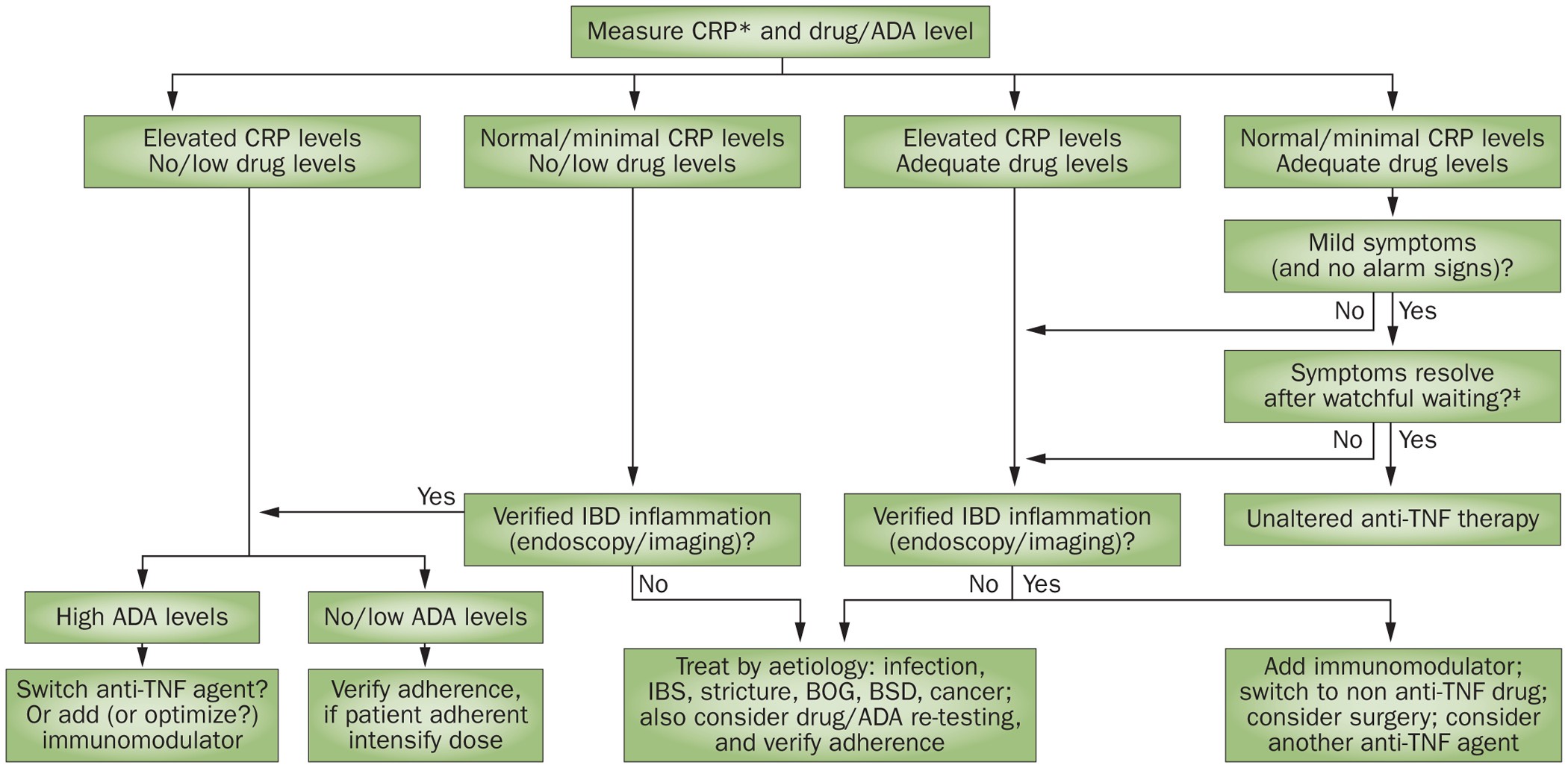
Tailoring anti-TNF therapy in IBD: drug levels and disease activity | Nature Reviews Gastroenterology & Hepatology
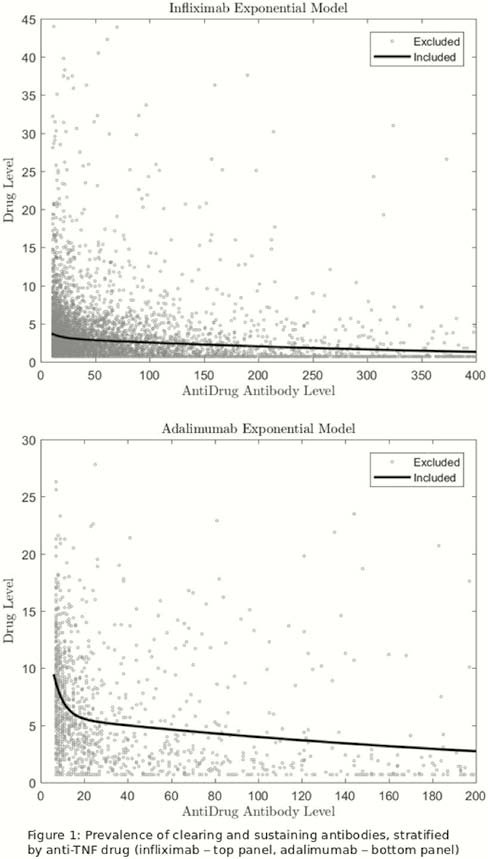
European Crohn´s and Colitis Organisation - ECCO - OP21 Positivity thresholds of total infliximab and adalimumab anti-drug antibody assay: The prevalence of clearing and transient anti-drug antibodies in a national therapeutic drug
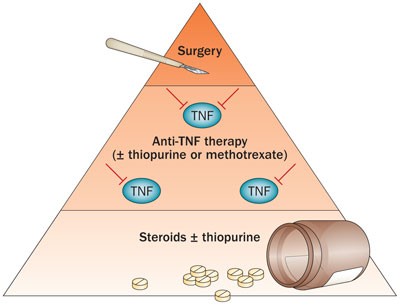
First-line therapy in adult Crohn's disease: who should receive anti-TNF agents? | Nature Reviews Gastroenterology & Hepatology

Dynamics of circulating TNF during adalimumab treatment using a drug-tolerant TNF assay | Science Translational Medicine

The Toronto Consensus Statements for the Management of Inflammatory Bowel Disease in Pregnancy - Gastroenterology

PDF) P553 Efficacy, safety and cost-efficiency of adalimumab 80 mg every other week in previously intensified IBD patients under treatment with adalimumab 40 mg every week
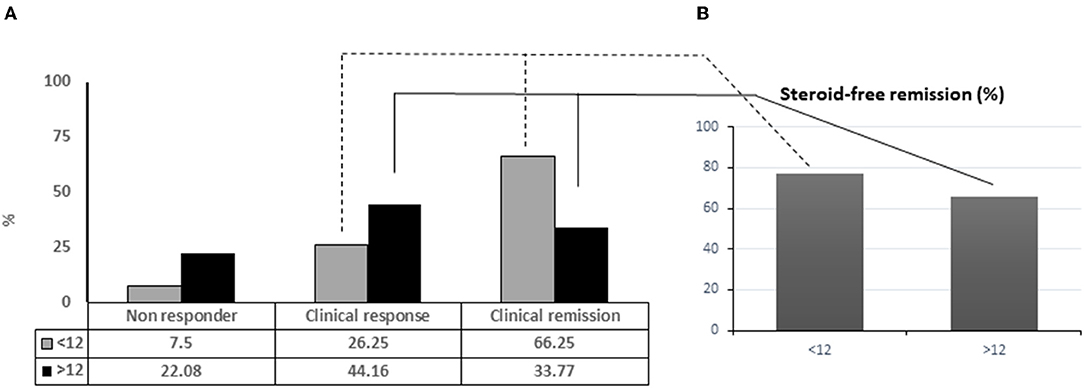
Frontiers | Administration Timing Is the Best Clinical Outcome Predictor for Adalimumab Administration in Crohn's Disease

Efficacy and safety of adalimumab in paediatric patients with moderate-to-severe ulcerative colitis (ENVISION I): a randomised, controlled, phase 3 study - The Lancet Gastroenterology & Hepatology

PDF) Efficacy and Safety of Adalimumab by Disease Duration: Analysis of Pooled Data From Crohn's Disease Studies

Increased versus conventional adalimumab dose interval for patients with Crohn's disease in stable remission (LADI): a pragmatic, open-label, non-inferiority, randomised controlled trial - The Lancet Gastroenterology & Hepatology

Biologics recommendations in the ECCO guidelines on therapeutics in Crohn's disease: medical treatment | Frontline Gastroenterology
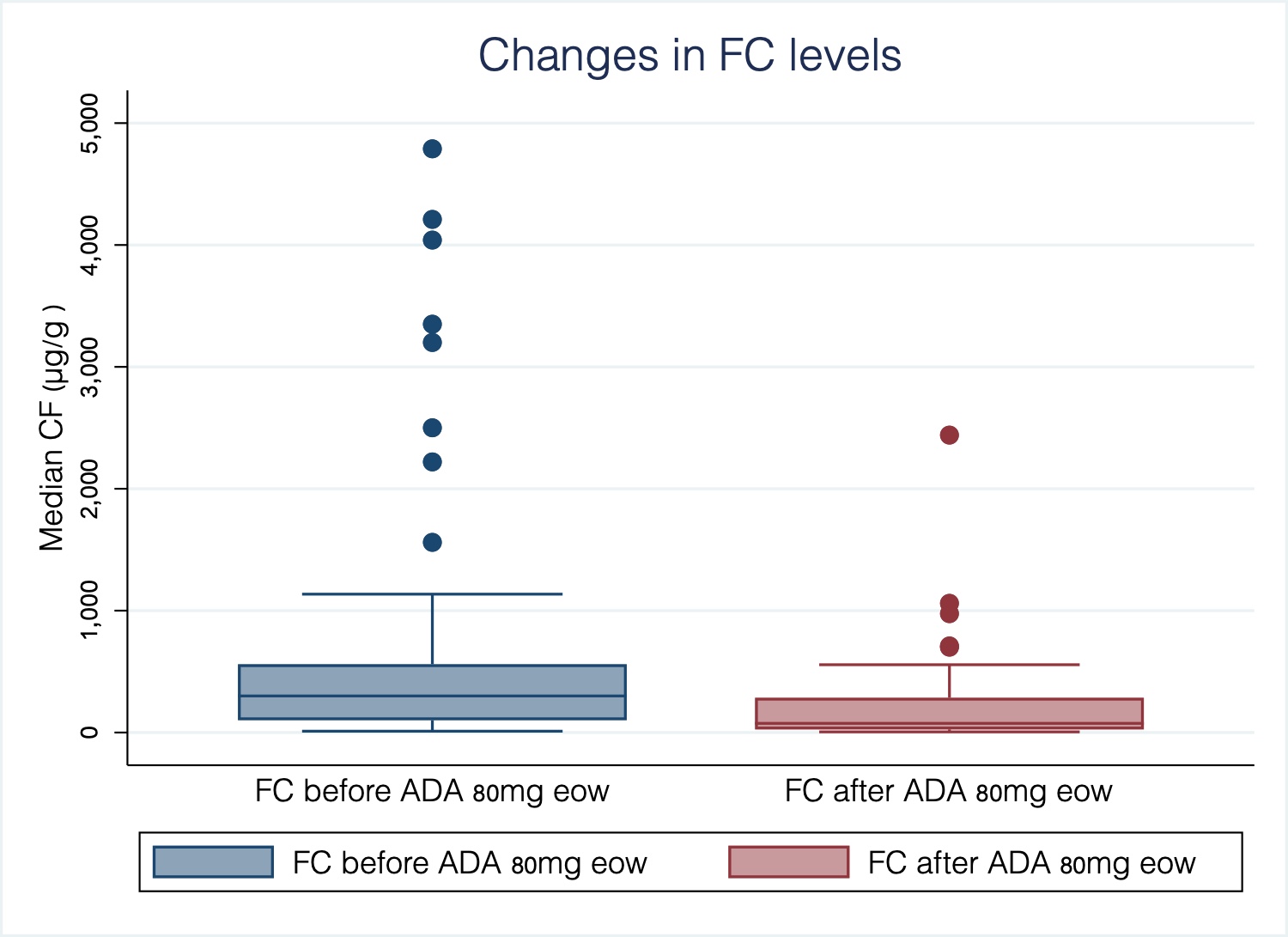
European Crohn´s and Colitis Organisation - ECCO - P533 Adalimumab 80mg every other week in inflammatory bowel disease: Treatment intensification outcomes in real life clinical practice

European Crohn´s and Colitis Organisation - ECCO - P533 Adalimumab 80mg every other week in inflammatory bowel disease: Treatment intensification outcomes in real life clinical practice